What we do
Combining our three core specialisms with cross sector experience from a world-class team, we transform businesses through the built environment and enhance the way people work.
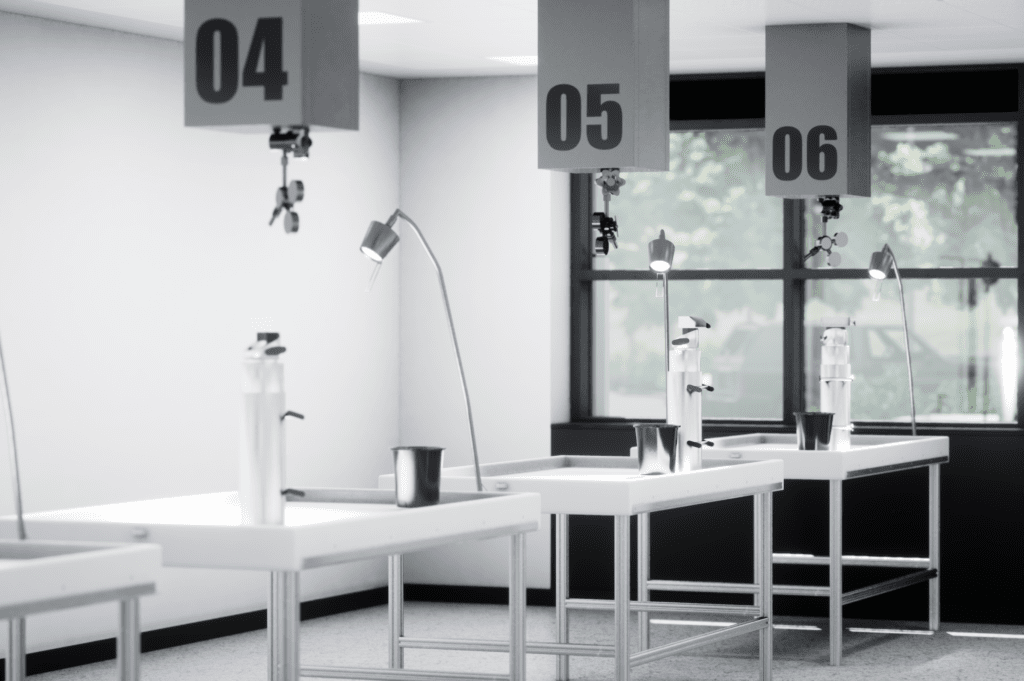
Find out what it’s like to go through a new laboratory build and design from a pharmaceutical testing lab owner.
Have you ever wondered about what it’s like to go through a new laboratory design and build? We interviewed the owner of a pharmaceutical testing lab to tell us about his experience of the process, from specification and design to the final fit out.
Check out the Q&A below to get a first-hand impression of what it’s like to design and build a new pharmaceutical laboratory.
AIS Life Sciences: What does your company do?
Lab owner: Our laboratory provides chemical and microbiology testing on medicinal products and raw materials, both active pharmaceutical ingredients (APIs) and excipients. We carry out testing on all formulation types, currently all our testing is non-sterile.
AIS: How does your industry effect your lab requirements?
LO: Lab requirements are impacted by both legislative regulations and by the types of clients that we work with. For example, regulations on testing for heavy metals have changed recently and different technology needs to be used to reach the required limits of detection. Safety is always a priority for all labs and this obviously effects the design of the lab and the equipment within it. For example, different chemicals require different filters for fume hoods and extraction units in the laboratory.
AIS: What requirements did you specify for your new lab?
LO: The lab needed to provide a secure entry/exit-controlled workspace and facilities for twenty people, including space for two fume hoods and a third if required. The laboratory space needed to be sub-divided into sample reception, sample preparation, physical lab, QC chemistry lab and microbiology lab. We also needed a separate meeting room and a lunch space.
AIS: What were the highest priorities for your new laboratory design?
LO: We wanted to future proof the lab, so a top priority was that it would give us space to grow the business by increasing our testing capacity. Our work is very instrument-driven so the number and placement of electrical sockets was also a priority. Grade A walls for the microbiology lab and ventilation to eight air cycles per minute were non-negotiable regulatory requirements too.
All this is set within the context of our workflow; we needed defined areas for sample reception, weighing and preparation, and chromatography. This was crucial as it set the scene for the day when analysts needed to know what areas they will be focused around.
AIS: What would you say were your team’s biggest priorities with the design?
LO: The main priority raised by the team was ensuring that we had sufficient electrical sockets to power our instruments. Particularly modelling how many staff can working a given area and planning the distribution of storage and instruments around them to maximise comfort and efficiency.
We also wanted to move from desktop PCs to laptops with monitors on wall mounts to free up bench space. Finally, colour was more important than we initially anticipated. The lab has to sell, opting for a clinical look and feel was something that would give our customers and clients the right first impression, as well as boost my team’s confidence.
AIS: Did you have a strict deadline, was the project time critical?
LO: The project was time critical as delays would have cost us revenue due to being unable to deliver work for clients. So we had a hard deadline of completion within a very specific time period and it was important to us to find a project partner that we felt we could trust with delivering this.
AIS: How did you factor the workflows of your new lab into its design?
LO: We are a GMP accredited laboratory so there is a requirement to ensure a controlled system and coherent unbroken end-to-end audit-trail from sample delivery to reporting of results. In the design of the lab, we considered sample delivery and in particular the log in room with a controlled secure entrance/exit and defined areas for logged in samples and quarantined samples. We kept the deign as simple as possible for both the physical movement of samples and the flow of information. For example, there is a route for documents to go directly from the sample delivery area to the office.
AIS: What internal factors did you consider when setting up your new laboratory? For example, branding and productivity?
LO: The vast majority of GMP labs look like classrooms – they’re boring, unattractive and uninspiring. We wanted to buck that trend and have a lab with a real ‘wow’ factor, that our employees would love! A shiny lab is an attractive lab and although our lab certainly isn’t huge it looks professional, modern and efficient which gives a safe trustworthy feel for our clients. The guys who work here are proud of their lab and look after it like it is home. Since opening, the feedback we have had from our staff and our clients has been overwhelmingly positive.
Other factors we considered were storage (you can never have enough!) and security.
AIS: What external factors were important to you when choosing your design?
LO: One of the main thing we were interested in was energy efficiency. Other than that, we also found it important to have lighting between tiles as it shows up dirt which is something that gets picked up in our audits.

AIS: Was your new design built around any specific pieces of capital equipment, or any laboratory processes?
LO: We knew the kit that we were planning to buy in the next five years so the design took that into account.
AIS: What are your overall thoughts on the fit-out process?
LO: The fit out went exactly as planned, agreed and costed for. The quality of the finish is really good and all the materials used are appropriate for a chemical lab.
AIS: How long did the new design and build take? Was this time frame expected?
LO: We planned for three months. The design and sign off took one month and from then on roughly another two months until final project completion on site so the project ran exactly to schedule
AIS: How did you account for social (workplace wellbeing) improvements in your design?
LO: Going through the design process has helped everyone here better understand how workspace layout impacts the physical and mental wellbeing of the people who work here. We’ll certainly be bearing this in mind with any future designs and expansions.
AIS: Are your laboratory team happy with the new lab?
LO: They love it! It’s clear the team enjoy working in the new lab and it has brought with it an unexpected boost to morale. New staff were able to get involved in the design process by contributing ideas which led to a strengthening of team culture and a real sense of unity.
Our team spend the majority of their time at work in the lab so it’s really important that they feel at home and comfortable in the lab facilities. Our clients also consistently tell us how impressed they are. It’s certainly done our credibility no harm at all!

AIS: What do you think was the most difficult part of the project?
LO: Making sure that we’d covered everything we needed in a way that was going to hit budget.
AIS: Is there anything you would do differently if you were to start the process again?
LO: (Laughs) get a bigger facility! I would also put more consideration into the electrical power supply to the unit.
AIS: What advice you would offer to those looking to build and design their own labs?
LO: Get references! Ask to see sites they have done and find people that have used them for feedback. Using the power of social media for this is a great way to see other case studies and how people felt about their final laboratory design outcome.
We hope this gives you some additional insight into the actual experience of the specification, design and build of a new lab and we would like to thank our interviewee for his time.
If you’re looking to design, fit out or refurbish a laboratory and workspace, we’ll be happy to discuss an initial strategy and your options. Get in touch to schedule a free no obligation consultation.